Home > Publications
Fan C, Fan M, Orlando BJ, Fastman NM, Zhang J, Xu Y, Chambers MG, Xu X, Perry K, Liao M, Feng L
doi: 10.1038/s41586-018-0330-9
Fan C, Fan M, Orlando BJ, Fastman NM, Zhang J, Xu Y, Chambers MG, Xu X, Perry K, Liao M, Feng L. X-ray and cryo-EM structures of the mitochondrial calcium uniporter. Nature. 2018 Jul;559(7715):575-579. doi: 10.1038/s41586-018-0330-9. Epub 2018 Jul 11. PMID: 29995856; PMCID: PMC6368340.
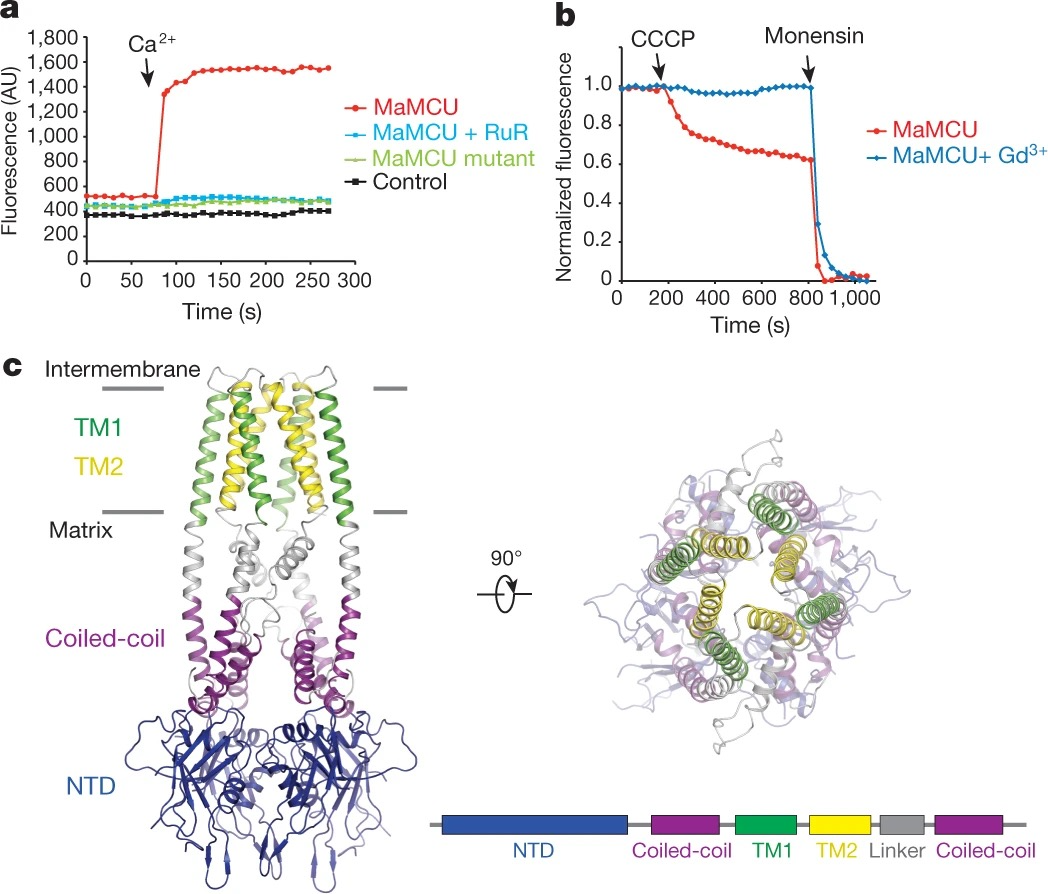
Mitochondrial calcium uptake is critical for regulating ATP production, intracellular calcium signalling, and cell death. This uptake is mediated by a highly selective calcium channel called the mitochondrial calcium uniporter (MCU). Here, we determined the structures of the pore-forming MCU proteins from two fungi by X-ray crystallography and single-particle cryo-electron microscopy. The stoichiometry, overall architecture, and individual subunit structure differed markedly from those described in the recent nuclear magnetic resonance structure of Caenorhabditis elegans MCU. We observed a dimer-of-dimer architecture across species and chemical environments, which was corroborated by biochemical experiments. Structural analyses and functional characterization uncovered the roles of key residues in the pore. These results reveal a new ion channel architecture, provide insights into calcium coordination, selectivity and conduction, and establish a structural framework for understanding the mechanism of mitochondrial calcium uniporter function.