Home > Publications
Thélot FA, Zhang W, Song K, Xu C, Huang J, Liao M
doi: 10.1126/science.abi9009
Thélot FA, Zhang W, Song K, Xu C, Huang J, Liao M. Distinct allosteric mechanisms of first-generation MsbA inhibitors. Science. 2021 Oct 29;374(6567):580-585. doi: 10.1126/science.abi9009. Epub 2021 Sep 23. PMID: 34554829; PMCID: PMC9109178.
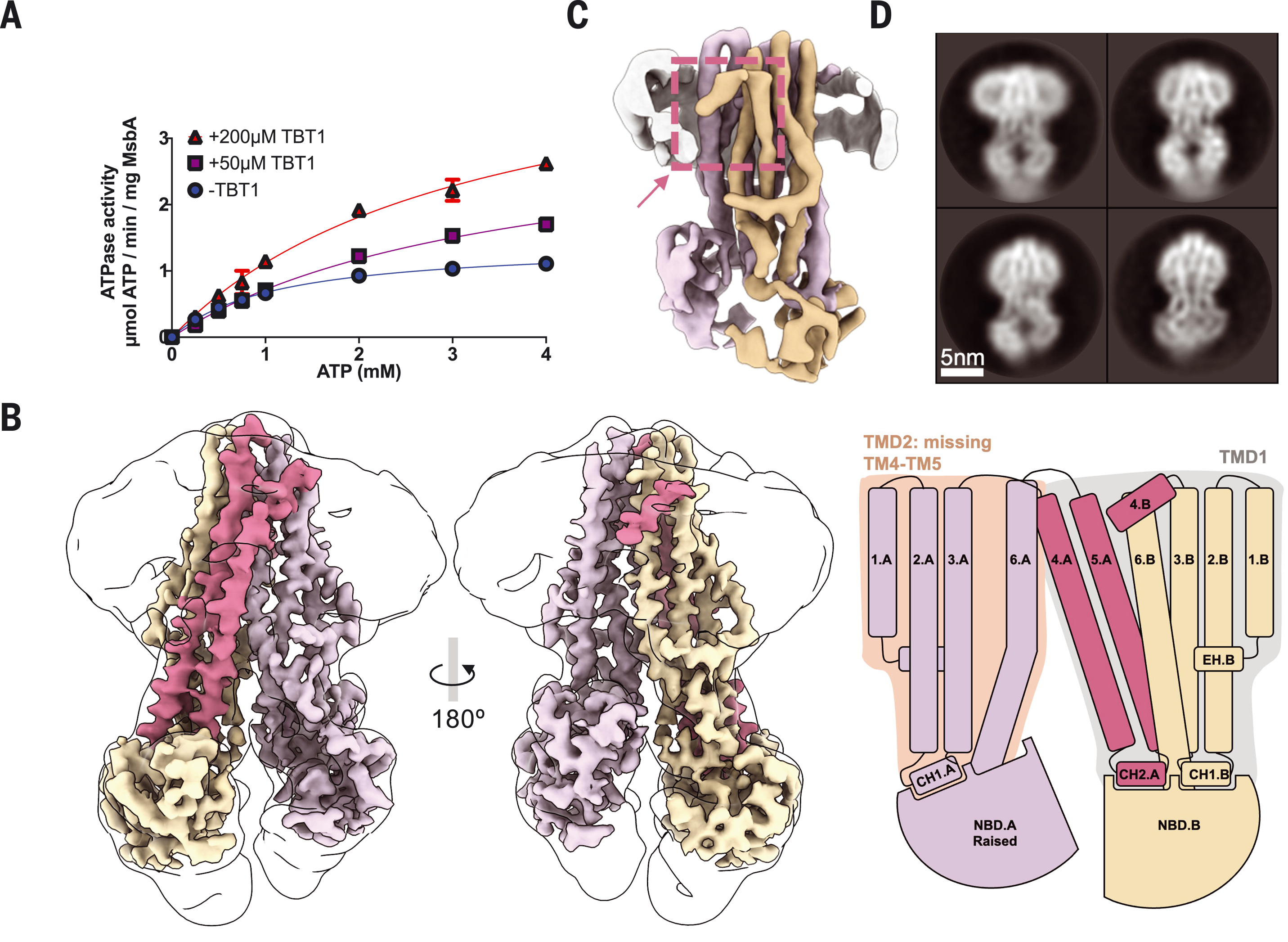
ATP-binding cassette (ABC) transporters couple adenosine 5′-triphosphate (ATP) hydrolysis to substrate transport across biological membranes. Although many are promising drug targets, their mechanisms of modulation by small-molecule inhibitors remain largely unknown. Two first-generation inhibitors of the MsbA transporter, tetrahydrobenzothiophene 1 (TBT1) and G247, induce opposite effects on ATP hydrolysis. Using single-particle cryo–electron microscopy and functional assays, we show that TBT1 and G247 bind adjacent yet separate pockets in the MsbA transmembrane domains. Two TBT1 molecules asymmetrically occupy the substrate-binding site, which leads to a collapsed inward-facing conformation with decreased distance between the nucleotide-binding domains (NBDs). By contrast, two G247 molecules symmetrically increase NBD distance in a wide inward-open state of MsbA. The divergent mechanisms of action of these MsbA inhibitors provide important insights into ABC transporter pharmacology.