Home > Publications
Sui X, Wang K, Gluchowski NL, Elliott SD, Liao M, Walther TC, Farese RV Jr
doi: 10.1038/s41586-020-2289-6
Sui X, Wang K, Gluchowski NL, Elliott SD, Liao M, Walther TC, Farese RV Jr. Structure and catalytic mechanism of a human triacylglycerol-synthesis enzyme. Nature. 2020 May;581(7808):323-328. doi: 10.1038/s41586-020-2289-6. Epub 2020 May 13. PMID: 32433611; PMCID: PMC7398557.
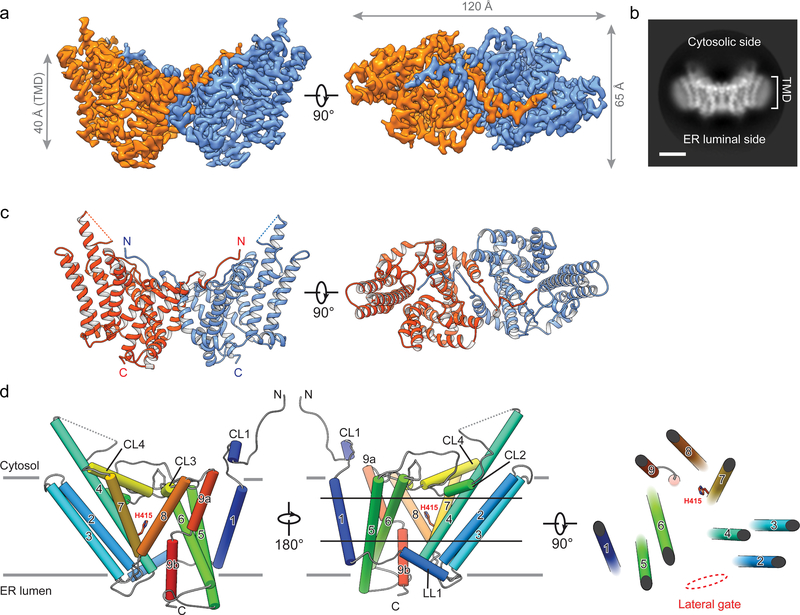
Triglycerides (triacylglycerols, TGs) store metabolic energy in organisms and have industrial uses for foods and fuels. Excessive accumulation of TGs in humans causes obesity and is associated with metabolic diseases. TG synthesis is catalyzed by acyl-CoA:diacylglycerol acyltransferase (DGAT) enzymes whose structures and catalytic mechanisms are unknown. Here we determined the structure of dimeric human DGAT1, a member of the membrane-bound O-acyltransferase (MBOAT) family, by cryo-electron microscopy at 3.0-Å resolution. DGAT1 forms a homodimer through N-terminal segments and a hydrophobic interface, with putative active sites within the membrane region. A structure obtained with oleoyl-CoA substrate resolved at 3.2-Å shows that the CoA moiety binds DGAT1 on the cytosolic side and the acyl group lies deep within a hydrophobic channel, positioning the acyl-CoA thioester bond near an invariant catalytic histidine residue. The reaction center is located inside a large cavity, which opens laterally to the membrane bilayer, providing lipid access to the active site. A lipid-like density, possibly an acyl-acceptor molecule, is located within the reaction center, orthogonal to acyl-CoA. Insights provided by the DGAT1 structures, together with mutagenesis and functional studies, give rise to a model of catalysis for DGAT’s generation of TGs.